By Dr. Susan E. Brown, PhD
Vitamin D and calcium are both vital nutrients for keeping your bones healthy and strong. However, there is a great deal of confusion about calcium and Vitamin D for bone health, especially when it comes to how much women really need. So, let’s take time to clear things up.

Much of the misunderstanding centers around calcium’s role in women’s bone health. Is calcium essential for healthy bones? Absolutely, but the mineral is not the “silver bullet” for bone health we’ve been mistakenly led to believe. As I will explain, calcium alone is not enough to strengthen bone; many other vitamins and minerals are essential to this process. Vitamin D, on the other hand, is often (and wrongly) downplayed by conventional medicine. As a result, you may be lacking adequate Vitamin D, putting your bones at risk. As you make decisions about how much Vitamin D and calcium supplementation you need, I want you to have the whole picture.
Table Of Contents
Why are calcium and Vitamin D important for bone development?
There’s a reason why I am including discussion of calcium and Vitamin D supplementation in the same article. Not only are these the two nutrients that I am asked about most frequently (see my FAQs below), but the two are highly connected when it comes to bone development. Vitamin D is the key to calcium absorption. To paraphrase noted Vitamin D researcher Dr. Michael Holick, you can swim in calcium and it will do no good unless you have adequate Vitamin D. Specifically, without Vitamin D your body can only absorb 10–15% of dietary calcium — so even if calcium is present, the body can’t use it! When Vitamin D is added, the absorption of dietary calcium increases to 30–40%. So, both calcium and Vitamin D are necessary to prevent osteoporosis — including postmenopausal osteoporosis. It all comes down to whether you are getting the right amounts of calcium and Vitamin D, as well as other key nutrients. Let’s explore this in greater detail.

What is Vitamin D and its role in the body?
Vitamin D is actually a precursor hormone — the building block of a powerful steroid hormone in your body called calcitriol. Direct sunlight triggers Vitamin D production in the skin and some foods provide beneficial amounts of Vitamin D, including wild caught salmon and some mushrooms. Vitamin D benefits bone health by working in concert with calcium, other nutrients and hormones in your body to support healthy bone renewal and reduce risk for osteoporosis. Researchers are discovering that Vitamin D also promotes normal cell growth and differentiation throughout the body.
Results of taking Vitamin D on a daily basis
- Less risk of hip fracture. Almost everyone who suffers from a low-trauma hip fracture has inadequate Vitamin D levels. In fact, one UK study found that 95% of those suffering hip fractures were deficient in Vitamin D. I believe that low Vitamin D is a major — and maybe even the most important — risk factor for hip fracture and osteoporosis.
- Improved heart health. Vitamin D helps maintain healthy blood pressure, suppress artery-damaging inflammation and protect the vessels from calcification. As a result, Vitamin D deficiency is inversely associated with heart attacks, strokes, high blood pressure and heart disease deaths. Pretty impressive when you consider that heart disease is our #1 cause of death!
- Cancer protection. Vitamin D is an excellent modulator of immune function, helping to protect you from infection and several common types of cancer.
- Diabetes prevention. Vitamin D helps prevent diabetes by enhancing insulin output and insulin sensitivity. People with low levels of Vitamin D have poorly functioning insulin-producing cells and an inadequate response to insulin, even when their blood sugar levels are normal.
In emerging research, Vitamin D has also been identified as helping to improve immune function and lessen the severity of Covid-19 symptoms.

What is calcium and its role in the body?
Calcium is the most abundant mineral in the human body and has several important functions. Two percent of our total body weight is made of calcium. And more than 99% of total body calcium is stored in the bones and teeth, where it supports their structure. When dietary calcium intake is insufficient and blood levels of calcium drop too low, the body will obtain calcium by extracting it from the bones. Ideally, calcium that is taken from the bones will be replaced when calcium levels are replenished. Milk and dairy can be a good source for calcium, especially yogurt, which also contains beneficial probiotics. There are many dairy-free choices too.
Good dairy-free food sources of dietary calcium include:
- Vegetables in the brassica family: broccoli, kale, cabbage (e.g., bok choy)
- Dark leafy greens, e.g., collards, spinach, turnip greens, dandelion greens, mustard greens, beet greens
- Rhubarb
- Canned fish/crustaceans with bones, e.g., sardines, pink salmon and shrimp
- Beans/legumes, e.g., edamame, tofu, tempeh, black-eyed peas and black beans
- Okra
- Nuts and seeds, e.g., almonds, sesame seeds
- Mineral water
- Herbal teas and infusions, e.g., oatstraw, nettle, red clover
Before you reach for that glass of mineral water or bowl of greens, however, you need to realize that to replace the calcium in your bones, an intricate process involving intake, metabolism and utilization must take place. While calcium is clearly important, there are at least 19 other key nutrients that each play a vital role in the structural integrity and overall health of our bones. To put the larger picture in context, I find it is sometimes useful to think of bone as a brick wall where the bricks are made of calcium and the other key nutrients make up the mortar. Without mortar, the wall is unstable. Bricks may fall out, making the wall even weaker. Just like a brick wall without mortar, research has shown that without Vitamin D, as well as Vitamin K2 and magnesium and other key nutrients, bone will lose its calcium.
How much calcium and Vitamin D should you take for osteoporosis?
Getting enough calcium for bone health
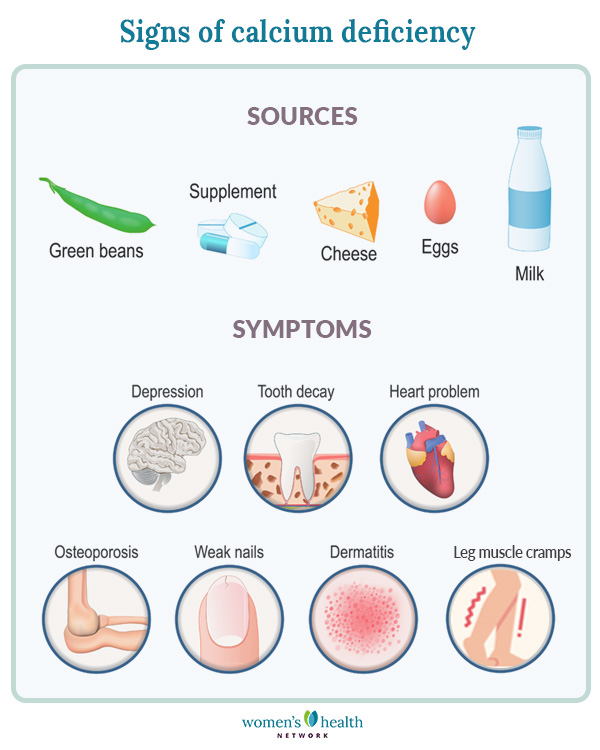
The body cannot produce its own calcium, so every day when we lose calcium through our skin, nails, hair, sweat, urine and feces, we need to replace it. It’s to your advantage to get your supplemental calcium through a high quality bone health supplement that comes complete with other nutrients needed for calcium absorption. The current recommended calcium intake for adult women up to age 51 is 1000 mg a day total between diet and supplements. Women ages 51-70 are recommended to take in 1200 mg a day. The good news is that most women are getting a good portion of their daily calcium through their diet. Therefore a beneficial therapeutic range for calcium supplementation is now generally 300 mg. Keep track of the foods you eat to get a sense of your typical dietary calcium intake and supplement to meet the optimal range. You may need more calcium if you don’t absorb it as well as most people. Nocturnal leg cramps, for instance, may indicate a higher need for calcium and greater attention to eating calcium-rich foods. See our graphic for even more symptoms of calcium deficiency.
Getting enough Vitamin D for bone health
Your body can’t create Vitamin D on its own. Instead, it’s designed to make it through sun exposure. In theory, you can make an ample supply of Vitamin D with as little as a couple of hours per week in the sun — provided the UVB rays are strong enough. You can also ingest D through food, especially fatty fish like wild–harvested salmon. Plus, lots of foods are fortified nowadays, so Vitamin D deficiency should be an easy problem to solve, right? But the truth is, most Americans just don’t get enough Vitamin D — especially during the winter months, when there is less sunlight. Certain other types of people may also be at a higher risk for Vitamin D deficiency.

The USDA recommends Vitamin D intake of between 400–800 IU/day, or 10–20 micrograms. The low end of 400 IU is simply too low for most of us to get any real benefit. Some studies have shown that adults need 3000–5000 IU per day. Others indicate healthy adults can readily metabolize up to 10,000 IU Vitamin D per day without harmful side effects. The European Union’s Scientific Committee on Food lists 2000 IU per day as the safety cut-off, as does the US Food and Nutrition Board. Can you get too much Vitamin D? Yes, but as we are learning, the amount needed to trigger toxicity is much more than what researchers are recommending for ideal supplementation range.
How much Vitamin D is best for you?
Today we know precisely what levels of Vitamin D in the blood are needed for optimal bone health: a minimum of 34 mg/mL, but more ideally, at least 50–60 mg/mL. Confirming the actual amount of Vitamin D in your blood lets you know if you need to take steps to increase levels of this key nutrient. I recommend every woman I work with to have her Vitamin D level tested using the 25(OH)D blood test. Testing is easy, and one of the most powerful tools there is to work with to achieve bone health. I will also note that the gold standard for Vitamin D supplementation is 2000 IU (50 mcg). On balance, 2000 IU of Vitamin D is a safe level that will give you enough of the vitamin to promote optimal bone health. This is the amount of Vitamin D that I include in my Better Bones Builder formula for these reasons. Your testing may reveal the need for additional supplementation, but 2000 IU daily can be a good starting point.
The best calcium and Vitamin D supplements
How to choose a calcium supplement for bone health and osteoporosis
Not all calcium supplements are created equal. Read ingredients labels carefully, checking for forms of calcium that are readily absorbed by the body. My suggestion is to look for a form of calcium which is well absorbed and highly alkalizing, such as seaweed-sourced calcium. Another big deciding factor is choosing supplements that balance calcium with other nutrients that research shows are critical for healthy metabolism of calcium. These might include magnesium (Cal-mag supplements), Vitamin K2 and Vitamin D. A multi-mineral/multi-vitamin supplement like my Better Bones Builder is formulated with exactly what you need for optimal absorption of calcium. If the only ingredient in the supplement is calcium, you will need to balance it out with other nutrients. This can be time-consuming and leave you open to error. Taking too much calcium without getting enough Vitamin K2, for example, could contribute to arterial calcium buildup, which you want to avoid. If you are not prepared to do the detective work on all these different supplements, stick with the ease of a high quality multi for bone health.
Vitamin D supplements for bone health and osteoporosis
Supplement your Vitamin D intake with the same form of Vitamin D that your skin produces naturally when exposed to the sun: Vitamin D3, or cholecalciferol. Make sure to avoid products with Vitamin D2 because your body cannot use it well. Supplementing is the best way to ensure you get enough Vitamin D each day — and it’s easy! The pills are small and easy to swallow, or you can use drops or even gummies. Vitamin D supplements are very inexpensive — but you still have to remember to take them every day.
Questions and answers about calcium and Vitamin D
Every day I’m asked questions like, “When should I take my calcium?” “Can I take too much Vitamin D?” “Can I get enough Vitamin D through sunlight?” and more. Here are some answers to these frequently asked questions.
Q: When should I take my calcium supplement?
It’s best to take calcium supplements with food so they absorb better. Ideally, spread them out over two meals for best absorption. Blood calcium can dip at night, so it helps to take some of your supplemental calcium with dinner.
Q: Can you take too much calcium?
Yes. Although we need calcium in relatively large quantities, you can take too much. For those looking to maximize bone health generally, supplement with calcium in the range of 300 mg/day. (The rest comes from a standard diet.) I don’t recommend using over 1000 mg supplemental calcium, as doctors tended to prescribe in the past.
Q: I get plenty of sunshine. Why do I need Vitamin D?
Even if you’re getting a lot of sunlight or taking supplements, you can still have low Vitamin D. That’s because each person’s body absorbs and uses Vitamin D differently, and the variation can be dramatic. As you go through life, your body may become less efficient at making, and using, Vitamin D for bone health. If you are getting enough direct sunlight (i.e., you live in the southern latitudes and get around 20 minutes of all over exposure a day), it’s still a good idea to get your Vitamin D level checked just to be sure.
Q: I’ve been reading a lot about Vitamin K2 and calcium. Should I be taking that too?
We have recently learned that Vitamin K2 is critical for calcium utilization and regulation. Vitamin K2 helps proteins bind to calcium and transports them to where they are needed in bones, organs and other tissues, and away from where excess calcium is not needed, including the arteries. It is also needed in the bone to produce a protein called osteocalcin. As the name suggests, osteocalcin helps bind calcium to the bone. Vitamin K2 as MK-7 offers additional benefits.









